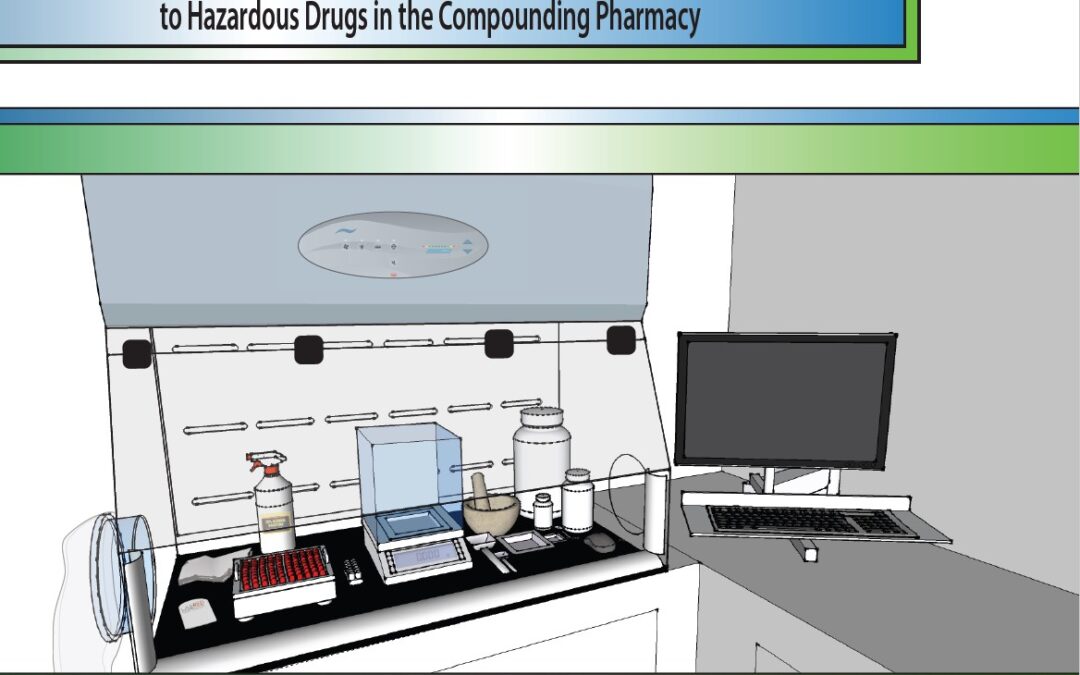
by Bryan Prince | Mar 10, 2022 | Uncategorized
Handling hazardous drugs in the compounding pharmacy, per USP chapter <800> guidelines, involves strategic workflow strategies with the goal of minimizing personnel and environmental exposure. In our previous article, “Can A Class I and Class II Biological...

by Bryan Prince | Dec 28, 2021 | Uncategorized
One of the most common issues a cleanroom will encounter is inconsistent temperature and humidity parameters. Because of the varying weather conditions and atmospheric pressure outside the building, conditions inside the building also fluctuate. As a very dry and...

by Bryan Prince | Sep 21, 2021 | Uncategorized
No two cleanroom suites are designed alike. Effective PEC placement will differ for sterile non-hazardous compounding versus sterile hazardous compounding, for example. Further, some cleanrooms may have space constraints, presenting challenges for placing PECs, fan...
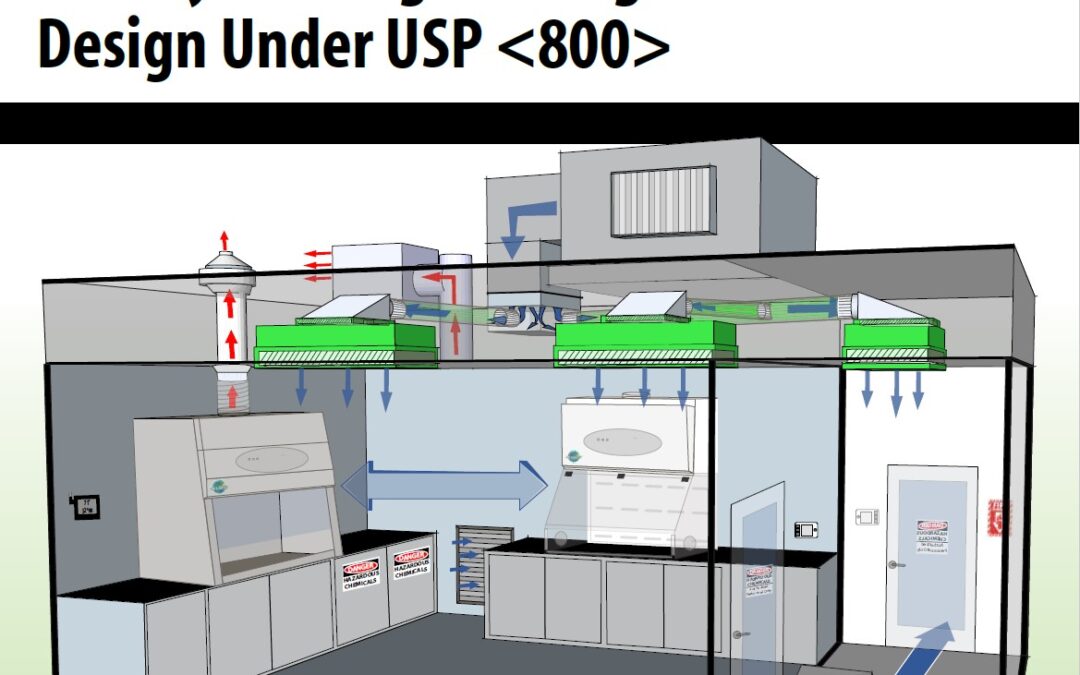
by Bryan Prince | Feb 16, 2021 | Uncategorized
In light of the recently published versions of USP chapters <795>, <797>, and <800>, with the intention of creating cohesive alignment between nonsterile and sterile compounding, it is essential to consider engineering controls for Hazardous Drug...
by Bryan Prince | Jan 16, 2021 | Uncategorized
Original Article Published Pharmacy Purchasing Products Magazine: Link to Full Article: Design a <795> Compliant Compounding Space : September 2019 – Pharmacy Purchasing & Products Magazine (pppmag.com) The purpose of USP General Chapter...





